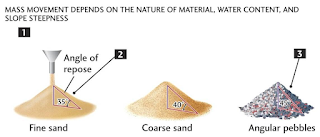
Aim: Determination of Angle of repose, Carre's Index & Hausner ratio of given powder Reference: L. Lachman CVS Subramanyam Requirements: Apparatus: Funnel, beaker, measuring cylinder, funnel holding stand Chemicals: Lactose, Na. carbonate, Boric acid Theory topics: Angle of Repose: Introduction, Definition, Equation, Description, various methods of determination, factors affect, Pharmacopoeia specification, effect on flow property Fig: Methods of angle of repose Carr’s consolidation Index (CI) Introduction, Definition, Equation, Description, various methods of determination, factors affect, Pharmacopoeia specification, effect on flow property Hausner’s Ratio: Introduction, Definition, Equation, Description, various methods of determination, factors affect, Pharmacopoeia specification, effect on flow property Procedure: Angle of repose: Weighed amt. of powder was taken (lactose-5gm, na. bicarbonate-10gm, Boric acid-10gm) Weighed p...


Nice Blog! Please Explore The Best Pharma Franchise And Third-party Manufacturing In India Leading Pharma Companies in Bhandara & Chandrapur, Maharashtra
ReplyDelete